20 Minute Labs: Make Your Own Geodes

MAKE YOUR OWN SPARKLING GEODES!
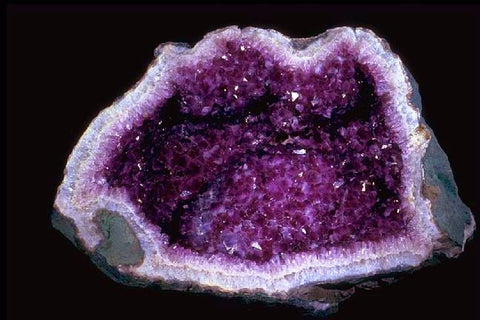
Have you ever seen a geode? On the outside, it looks like a plain old, round rock. But when you break it open, inside is a lining of beautiful crystals!
Geodes are found all over the world, usually in deserts. If you live in any of the southwest or midwestern states, you might be able to find them out in nature!
You can also find them for sale at toy and rock shops to take home and break open yourself.
Geodes start out as tree roots, mud balls, or volcanic bubbles inside of sedimentary rock. Over many millions of years, the outer, rounded shell hardens, but the inside remains hollow.A liquid containing dissolved minerals gathers on the inner walls of the geode. Over time, these minerals form crystals!
 In this experiment, you can create some geode-like crystals using eggshells and a few items from home. The science is similar to how a real geode is made, but on a shorter time scale - 20 minutes instead of millions of years!
In this experiment, you can create some geode-like crystals using eggshells and a few items from home. The science is similar to how a real geode is made, but on a shorter time scale - 20 minutes instead of millions of years!
NOTE: Setting up the lab will only take about 20 minutes, but once you're done you'll have to wait overnight to see the full results.
What You'll Need
-
3 eggshell halves
- measuring cup
- 4 cups of water
- pot
- 1 3/4 cups borax
- spoon
- 3 glasses with lids
- food coloring
Let's Get Started!
-
 Collect eggshell halves. You can either hard-boil eggs and peel off the shells or crack open raw eggs and wash and dry the shells before using.
Collect eggshell halves. You can either hard-boil eggs and peel off the shells or crack open raw eggs and wash and dry the shells before using. - With an adult's help, bring four cups of water to a boil in a pot on the stove.
- Remove the pot from the heat and SLOWLY stir in 1 3/4 cups borax until completely dissolved. (NOTE: Avoid splashing; do not inhale fumes; turn on air vent and/or open windows.)
- Let the solution cool for about 15 minutes.
- To each of the three glasses, add about 10 drops of food coloring. We found that lighter colors, like yellow, work well.
-
Carefully pour the borax solution into the three glasses so there is about the same amount in each one. The food coloring should easily go into solution.
- Now add an eggshell to each glass, allowing it to sink to the bottom. Use a spoon to carefully arrange the shell so it isn't touching the sides of the glass to prevent sticking.
- Cover each glass with a lid. (This will prevent borax fumes from escaping.)
- Leave the glasses undisturbed overnight.
- The next day, carefully then scoop out the eggshell with a spoon. It will likely be stuck to the bottom of the glass, so you may have to use a bit of pressure to release it.
- Place on a plate or paper towel to dry. Enjoy your geode!
WHAT'S GOING ON?
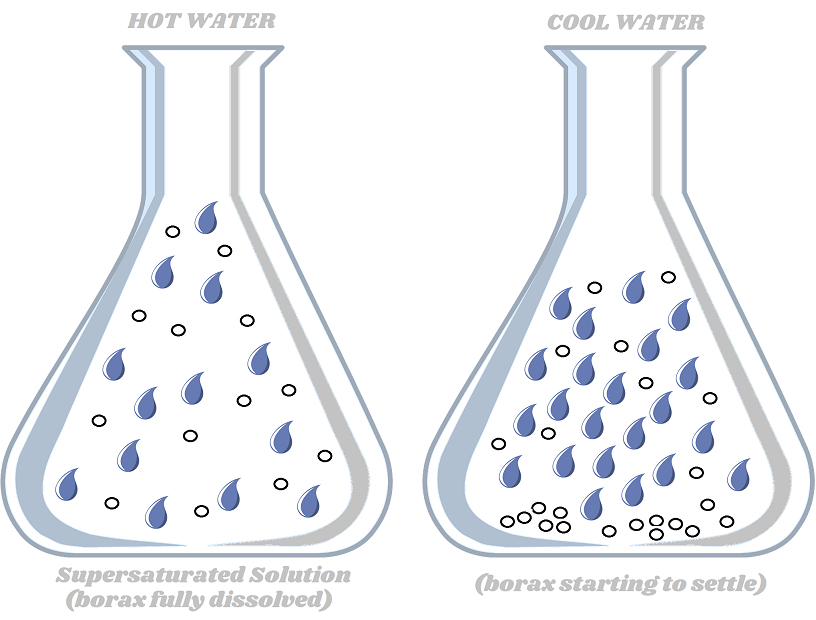 You probably noticed that the borax powder dissolved easily in the hot water. More borax will dissolve in hot water than in cold water. Hot water molecules move around a lot more and there is more space between the molecules. So there is more space to hold the borax. This type of solution is called a supersaturated solution. The water can not hold any more borax - it's completely stuffed!
You probably noticed that the borax powder dissolved easily in the hot water. More borax will dissolve in hot water than in cold water. Hot water molecules move around a lot more and there is more space between the molecules. So there is more space to hold the borax. This type of solution is called a supersaturated solution. The water can not hold any more borax - it's completely stuffed!
When the solution cooled overnight, the water molecules slowed down and got closer together. There was no more room for the borax, so it could no longer remain dissolved. The borax particles came out of solution and settled on the eggshells, in the form of crystals!
What is a Crystal?
 When molecules like borax (a mineral salt) come together in a very organized, patterned way, a crystal is formed. The size and shape of the crystals depends on how fast the solution cools. When solutions cool fast, smaller crystals are formed because they have less time to organize. Slow-cooling solutions tend to form larger crystals. You might want to repeat the experiment again, but this time, try putting the glasses into the refrigerator overnight. What do you think will happen to the size of the crystals?
When molecules like borax (a mineral salt) come together in a very organized, patterned way, a crystal is formed. The size and shape of the crystals depends on how fast the solution cools. When solutions cool fast, smaller crystals are formed because they have less time to organize. Slow-cooling solutions tend to form larger crystals. You might want to repeat the experiment again, but this time, try putting the glasses into the refrigerator overnight. What do you think will happen to the size of the crystals?
SHARE WITH US!
Let us know how your experiments turned out! Share your photos and results with us on Facebook, Twitter, Instagram, or send us an email to info@yellow-scope.com. We love getting your messages!
For more exciting experiments, check out our Yellow Scope science kits on the Shop tab of our website!


 3 eggshell halves
3 eggshell halves Collect eggshell halves. You can either hard-boil eggs and peel off the shells or crack open raw eggs and wash and dry the shells before using.
Collect eggshell halves. You can either hard-boil eggs and peel off the shells or crack open raw eggs and wash and dry the shells before using. Carefully pour the borax solution into the three glasses so there is about the same amount in each one. The food coloring should easily go into solution.
Carefully pour the borax solution into the three glasses so there is about the same amount in each one. The food coloring should easily go into solution. 


Chelsea Schuyler
Author